Abstract
Background: Chronic myeloid leukemia (CML) is a myeloproliferative neoplasm that typically occurs in the fifth and sixth decades of life with presentations usually confined to hematopoietic tissues—primarily blood, bone marrow, and spleen. However, primary presentation of CML as extramedullary involvement (osteolytic lesion) is extremely rare, occurring in less than 1% of cases.
Case Presentation: Here, we present a case of a 25-year-old male with chronic myeloid leukemia (CML), characterized by an atypical presentation of a femur fracture accompanied by an osteolytic lesion. The peripheral blood findings displayed typical features of CML, including leukocytosis with a full spectrum of myeloid maturation stages, basophilia, and occasional blasts. The diagnosis of CML in the blastic phase was confirmed by the presence of the Philadelphia chromosome and the BCR-ABL1 fusion gene (b2a2), as well as features of myeloid sarcoma in the tissue biopsy.
Conclusion: Given the aggressive nature of granulocytic sarcoma and its impact on prognosis, early recognition and intervention are essential to mitigate complications and improve patient outcomes.
Introduction
Chronic myeloid leukemia (CML) is a Philadelphia-positive myeloproliferative neoplasm that typically presents at a median age of 50 to 60 years, with the majority presenting in the chronic phase, characterized by blasts less than 5% and the absence of extramedullary involvement1. According to Q Lin et al., the incidence of CML varies considerably worldwide, with rates ranging from 0.4 to 1.75 cases per 100,000 people in different regions. A study conducted in East Malaysia pinpointed a particularly high estimated prevalence and incidence of CML in southern Sarawak, standing at 69.2 cases per 1,000,000 population2, 3. Furthermore, extramedullary manifestations of the disease are noted to be extremely rare in this context.
Its presentation primarily involves hematopoietic tissues, including blood, bone marrow, liver, and spleen. Based on the most recent research conducted by Terjanian et al., extramedullary manifestations of chronic myeloid leukemia are exceptionally uncommon. The primary sites frequently involved include lymph nodes, bones, and skin. Bone lesions are characterized by some as osteoblastic, others as osteolytic, and in rare instances, they may present with both features simultaneously. Extramedullary disease indicates a blast crisis and is associated with a poor prognosis. According to the findings from Terjanian's study, out of the 24 patients analyzed, a staggering 96% progressed and eventually succumbed to the disease4.
This report underscores an unusual instance of CML in a young adult who suffered a rare femur fracture accompanied by an osteolytic lesion. Such an event has been documented in only a handful of cases before, making it an exceedingly rare occurrence. The objective of this case study is to raise awareness among orthopedic surgeons to consider granulocytic sarcoma as a potential diagnosis for hip pain. Prompt diagnosis and early intervention are essential in instances of this atypical CML presentation because the prognosis is expected to be unfavorable due to the development of complications, as illustrated in the case we have presented.
| Parameter | Result | Normal reference range |
| White blood cell count (x10 9 /L) | 64 | 4-11 |
| Haemoglobin (g/dL) | 12.4 | 13.5-17.6 |
| Platelet (x10 9 /L) | 646 | 150-450 |
| Peripheral blood film report | Hyperleucocytosis with presence of all the stages of myeloid maturation, basophilia, occasional blasts and thrombocytosis with platelet anisocytosis | |
| Bone marrow aspirate and biopsy | Increased cellularity due to granulocytic proliferation and supported the diagnosis of myeloproliferative neoplasm | |
| Cytogenetic study | 46, XX, t(9;22) (q34;q11) | |
| Molecular analysis | Major BCR-ABL1 fusion gene (b2a2) | |
CASE PRESENTATION
Patient History
A 25-year-old Malay man with no known medical history presented with right hip pain and difficulty walking following a fall one day prior to his presentation. There was no history of bone pain, swelling, or any constitutional symptoms. Physical examination revealed tenderness in the upper region of the right thigh and restricted range of motion in the right hip joint. There were no bruises, swelling, or skin changes, and no remarkable findings in the other systems. There was neither lymphadenopathy nor hepatomegaly, but mild splenomegaly was noted (dull Traube's space).
Diagnostic Findings
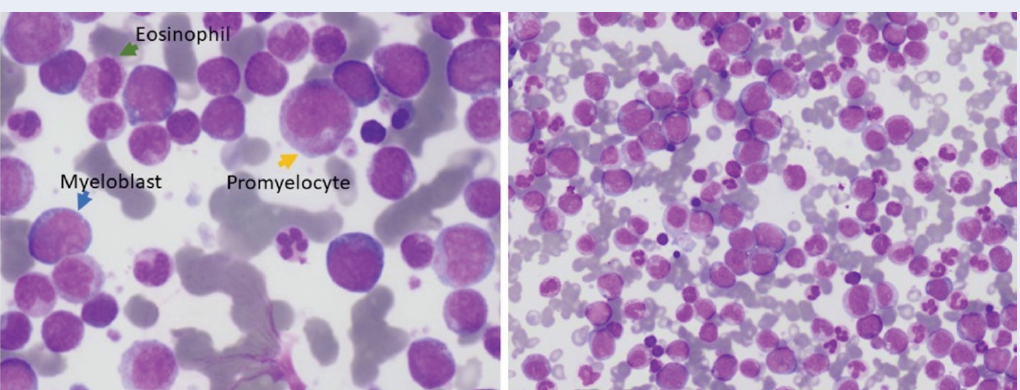
The initial X-ray of the pelvis showed a fracture accompanied by a lytic lesion extending from the greater to the lesser trochanter of the right femur. Subsequent magnetic resonance imaging (MRI) of the entire body revealed multiple lytic lesions at the greater trochanter of the right femur, skull, and lumbar vertebra (L5), with no indications of bone metastasis. Given the presence of multiple bony lesions, several potential diagnoses were considered, including Ewing sarcoma, osteosarcoma, and underlying metastasis. Due to observed leukocytosis, an urgent blood film was requested. The results of both the peripheral blood and bone marrow examination at the time of diagnosis are outlined in Table 1 and indicate chronic myeloid leukemia in the chronic phase, with blasts constituting less than 5% (refer to Figure 1).
The results of the bone and tissue biopsy from the right femur confirmed the presence of myeloid sarcoma (see Figure 2). Consequently, the diagnosis was revised to CML in the blast phase. The patient underwent a marginal resection and the placement of a proximal femur endoprosthesis for the fracture.
Treatment
Initially, the patient was administered the tyrosine kinase inhibitor (TKI) Imatinib (Glivec) at a daily dosage of 400mg, achieving a good hematological response within one month of initiating the TKI treatment. Additionally, the patient underwent chemotherapy using the AML 3+7 protocol, which involves administering daunorubicin for three consecutive days followed by cytarabine for seven days. No remission was documented during the treatment due to the suboptimal quality of the marrow obtained. This treatment approach was selected due to the presence of extramedullary disease indicating a blast crisis, and the patient was managed accordingly.
Outcome
However, after five months of treatment, a dislocation of the endoprosthesis occurred, resulting in the formation of a mass at the fracture site that subsequently became infected. Unfortunately, the patient's condition deteriorated, leading to his death due to septic shock resulting from neutropenic sepsis and disseminated intravascular coagulopathy.
DISCUSSION
Skeletal symptoms as an initial presentation of extramedullary involvement in CML are infrequent and are generally associated with acute blastic transformation. Radiographic evidence has revealed skeletal lesions in 16% of CML patients, encompassing diffuse osteoporosis, focal osteoblastic/osteolytic lesions, granulocytic sarcoma (also known as chloroma), and arthritis5. Radiologically, the differential diagnosis in adults includes non-hematological diseases such as metastasis, chondrosarcoma, or malignant fibrous histiocytoma6.
The osteolytic bony lesions observed in CML patients are postulated to arise from various mechanisms. One explanation suggests the direct invasion of leukemic cells into the bone, resulting in bone destruction. Another possibility is that leukemic cells trigger bone resorption, leading to localized bone destruction. Additionally, it is hypothesized that parathyroid hormone-related protein, produced by leukemic cells, acts on the PTH receptor, thereby stimulating bone resorption. These proposed mechanisms highlight the multifaceted nature of osteolytic lesions in CML and underscore the complexity of their pathogenesis7.
| Age and gender | 35-year-old male | 18-year-old male | 58-year-old male | 38-years-old male | 17-year-old female | 48-year old , gender not available |
| Chief complained | Fever and intense pain in both knees and ankles persisting for 5 days | Pain over the left shoulder over the past 4days | acute confusion, polyuria, and polydipsia for 7 days | Left buttock pain | Left hip joint pain | Right knee pain with reduced range of motion |
| Initial diagnosis | CML in CP on Imatinib | CML in CP on Imatinib | CML in CP | CML in CP on Imatinib | Myeloid sarcoma | Myeloid Sarcoma |
| Time elapsed from the initial diagnosis and extramedullary involvement (in months). | 12 months | 60 months | 72 months | 96 months | At presentation | At presentation |
| Treatment received | AML Induction therapy 7 + 3 and Dasatinib 140mg OD | Nilotinib 200mg BD | Received ponatinib and radiotherapy. | Received salvage combined chemotherapy | Dasatinib 70 mg BD and allo-HSCT after 4 months of the diagnosis | Imatinib. |
| Outcome | Discharged well till reported date. | Discharged well till reported date. | Expired 18 months after presentation | Expired | complete remission and achieved treatment free remission 30 months post-transplantation | Discharged well till reported date. |
| References | 8 | 9 | 7 | 10 | 11 | 12 |
To date, over the past five years, we have identified several cases of CML with osteolytic lesions (Table 2 ). Among these cases, two initially presented with osteolytic lesions. Two cases progressed to death, while one achieved treatment-free remission following a stem cell transplant. Based on the reviewed cases, it is noteworthy that patients with myeloid sarcoma who undergo allogeneic stem cell transplantation may benefit.
In scenarios akin to this, the disease is often not expected based on clinical observations, underscoring the need for a heightened level of suspicion. The blood and bone marrow characteristics conformed to the criteria for chronic phase chronic myeloid leukemia (CML). However, a tissue biopsy uncovered features suggestive of an unifocal extramedullary granulocytic sarcoma. The presence of extramedullary myeloblast proliferation classifies the CML as being in the blastic phase, necessitating swift diagnosis and aggressive treatment.
An accumulation of leukemic granulocytic precursors extramedullary, known as granulocytic sarcoma or chloroma, represents an infrequent and aggressive type of malignancy. Diagnosis can be challenging since it is not typically suspected during the initial presentation. Radiological assessments play a crucial role, and confirmation is achieved through histological examination. In terms of radiological presentations, skeletal lesions often manifest as lytic lesions, similar to the case at hand, necessitating differentiation from metastasis, malignant fibrous histiocytoma, or lytic chondrosarcoma6. Histologically, the diagnosis relies on the observation of sheets of leukemic granulocytic precursors or myeloblasts, confirmed by CD177 and MPO positivity.
According to the literature review by Soni et al. and the CML 2022 update by Jabbour et al., extramedullary manifestations pose ongoing challenges in the management of CML. The blast crisis represents a significant hurdle in CML management, emphasizing the need for early consideration of allogeneic stem cell transplantation following initial therapy with tyrosine kinase inhibitors (TKIs). Newer-generation TKIs such as dasatinib or ponatinib are preferred over imatinib to effectively reduce the CML burden. In cases where TKIs alone are insufficient, initiating acute leukemia-type induction therapy, such as FLAG-IDA or a combination of chemotherapy tailored to the immunophenotype, following the MD Anderson approach, may be warranted13, 14. This comprehensive approach highlights the importance of personalized treatment strategies to address the complexities of CML management.
CONCLUSION
The initial presentation of extramedullary involvement in CML patients is acknowledged to be exceedingly rare. The current case report seeks to raise awareness among general orthopedic surgeons regarding the possibility of granulocytic sarcoma in their differential diagnoses for hip pain. Reinforcing the importance of considering CML in the differential diagnosis of unexplained osteolytic lesions, especially in atypical patient populations, underscores the necessity for heightened awareness among clinicians beyond hematologists and oncologists. This includes orthopedic surgeons and emphasizes the need for future research in this area.
Abbreviations
AML: Acute Myeloid Leukemia, BCR-ABL1: Breakpoint Cluster Region-Abelson Murine Leukemia Viral Oncogene Homolog 1, CML: Chronic Myeloid Leukemia, MRI: Magnetic Resonance Imaging, PTH: Parathyroid Hormone, TKI: Tyrosine Kinase Inhibitor
Acknowledgments
None.
Author’s contributions
The initial concept is by Mohd Nazri Hassan and Zefarina Zulkafli. The first draft is written by Razan Hayati Zulkeflee, Mohd Nazri Hassan and Marini Ramli. Shafini Mohamed Yusoff and Norzaliana Zawawi contributed to the full blood investigation and biopsy results. Azlan Husin and Abu Dzarr Abdullah contributed to the management of this case. Rosnah Bahar and Wan Suriana Wan Abd Rahman are thoroughly reviewing it for essential intellectual substance. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Competing interests
The authors declare that they have no competing interests.
References
-
Khoury
J.D.,
Solary
E.,
Abla
O.,
Akkari
Y.,
Alaggio
R.,
Apperley
J.F.,
The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Vol. 36, Leukemia. Springer Nature; 2022. p. 1703–19. . 2022
.
-
Lin
Q.,
Mao
L.,
Shao
L.,
Zhu
L.,
Han
Q.,
Zhu
H.,
Global, Regional, and National Burden of Chronic Myeloid Leukemia, 1990-2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Frontiers in Oncology.
2020;
10
.
View Article PubMed Google Scholar -
Kuan
J. Win,
Michael
M. Sudau,
The epidemiology of chronic myeloid leukaemia in southern Sarawak, Borneo Island. .
.
-
Terjanian
T.,
Kantarjian
H.,
Keating
M.,
Talpaz
M.,
McCredie
K.,
Freireich
E.J.,
Clinical and prognostic features of patients with Philadelphia chromosome-positive chronic myelogenous leukemia and extramedullary disease. Cancer.
1987;
59
(2)
:
297-300
.
View Article PubMed Google Scholar -
Palejwala
A.H.,
O'Connor
K.P.,
Shi
H.,
Villeneuve
L.,
Scordino
T.,
Glenn
C.A.,
Chronic myeloid leukemia manifested as myeloid sarcoma: review of literature and case report. Journal of Clinical Neuroscience.
2019;
64
:
269-76
.
View Article PubMed Google Scholar -
Maloisel
F.,
Favre
G.,
Mahmal
L.,
Zamfir
A.,
Andrès
E.,
Isolated lytic bone lesion as extramedullary disease in chronic myelogenous leukemia: a report of three new cases. European Journal of Internal Medicine.
2005;
16
(4)
:
288-90
.
View Article PubMed Google Scholar -
Toro-Tobón
D.,
Agosto
S.,
Ahmadi
S.,
Koops
M.,
Bruder
J.M.,
Chronic myeloid leukemia associated hypercalcemia: A case report and literature review. The American Journal of Case Reports.
2017;
18
:
203-7
.
View Article PubMed Google Scholar -
Rujirachun
P.,
Junyavoraluk
A.,
Owattanapanich
W.,
Suvannarerg
V.,
Sirinvaravong
S.,
Journal of Medical Case Reports.
Leukemic arthritis and severe hypercalcemia in a man with chronic myeloid leukemia: A case report and review of the literature. Journal of Medical Case Reports.
2018;
12
:
1-7
.
View Article Google Scholar -
Gauthier
M.,
Mouchel
P.L.,
Syrykh
C.,
Couture
G.,
Chronic myeloid leukemia revealed through focal bone lesions responding to imatinib: the bee's knees. Joint, Bone, Spine.
2021;
88
(2)
.
View Article PubMed Google Scholar -
Tsukamoto
S.,
Ota
S.,
Ohwada
C.,
Takeda
Y.,
Takeuchi
M.,
Sakaida
E.,
Extramedullary blast crisis of chronic myelogenous leukemia as an initial presentation. Leukemia Research Reports.
2013;
2
(2)
:
67-9
.
View Article PubMed Google Scholar -
Wang
Y.,
Hospital
R.,
Tong
S.J.,
Sun
H.,
Zhang
S.,
Myeloid Sarcoma Transformed From CML Presenting Ectopic Expression of AFP: A Case Report. researchsquare.
2022;
2022
.
View Article Google Scholar -
Aisyi
M.,
Syarif
A.H.,
Meisita
A.,
Kosasih
A.,
Basuki
A.,
Sari
R.M.,
Pathological Fracture: An Unusual Presentation in Childhood Chronic Myeloid Leukemia. Indonesian Journal of Cancer..
2019;
13
(4)
:
140
.
View Article Google Scholar -
Jabbour
E.,
Kantarjian
H.,
Chronic myeloid leukemia: 2022 update on diagnosis, therapy, and monitoring. American Journal of Hematology.
2022;
97
(9)
:
1236-56
.
View Article PubMed Google Scholar -
Soni
A.,
Paluri
R.,
Deal
T.,
Peker
D.,
Extramedullary Involvement by Chronic Myelogeneous Leukemia in Five Patients With Unusual Clinicopathologic Features: A Review of the Effectiveness of Tyrosine Kinase Inhibitors. Journal of Clinical Medicine Research.
2016;
8
(6)
:
480-5
.
View Article PubMed Google Scholar
Comments
Article Details
Volume & Issue : Vol 11 No 3 (2024)
Page No.: 6276-6281
Published on: 2024-03-31
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
- HTML viewed - 3485 times
- PDF downloaded - 925 times
- XML downloaded - 110 times
 Biomedpress
Biomedpress




