Abstract
Introduction: Sirtuin 1 (SIRT1) emerges as a promising biomarker for heart remodeling in the context of essential arterial hypertension (EAH). Additionally, the levels of certain plasma peptides might signal myocardial fibrosis and the progression of heart failure (HF) in hypertensive patients. Despite this potential, conflicting data exist regarding the marker's diagnostic value in HF patients. This study aims to explore the impact of anthropometric factors on plasma SIRT1 levels in men diagnosed with EAH and HF, considering the rs7069102 single nucleotide polymorphism (SNP) in the SIRT1 gene.
Methods: The study included an examination of 190 Ukrainian men aged between 40 and 65. The participants were divided into two groups: a control group, consisting of 70 individuals without cardiovascular disease (CVD), and a study group comprising 120 men with EAH, 60 of whom displayed signs of HF. Plasma SIRT1 levels were quantified using an enzyme-linked immunosorbent assay (ELISA), while the rs7069102 C/G polymorphism in the SIRT1 gene was detected through allele-specific polymerase chain reaction (PCR). The research employed various statistical methods, including correlation analysis, T-test, Mann-Whitney U test, one-way ANOVA, and analysis of contingency tables for data analysis.
Results: The investigation revealed that men suffering from EAH and HF exhibited significantly reduced plasma SIRT1 levels (1.550 ± 0.084 ng/ml) compared to those with EAH but without HF (3.271 ± 0.238 ng/ml, p < 0.001). Notably, hypertensive men with concurrent HF and obesity or those with an early onset of hypertension showed even lower plasma SIRT1 concentrations. Interestingly, the analysis found no significant difference in plasma SIRT1 levels among individuals in the control group and EAH patients without HF across different rs7069102 C/G SNP variants. However, among hypertensive men with HF, individuals with the GG genotype displayed considerably lower plasma peptide levels compared to those with either the CC or CG genotype (1.390 ± 0.092 ng/ml vs. 1.744 ± 0.126 ng/ml, p = 0.032).
Conclusion: This study highlights a significant association between reduced plasma SIRT1 levels and heart failure in male patients with EAH. Factors contributing to decreased plasma peptide levels include obesity, early onset of hypertension, and possessing the GG variant of the rs7069102 SNP in the SIRT1 gene. These findings underscore the potential of SIRT1 as a marker for HF and may guide future therapeutic strategies.
Introduction
Currently, there's a concerning trend in the global increase of heart failure (HF) and associated mortality rates. For instance, approximately 6.7 million Americans are afflicted with HF. This number is predicted to rise, with the lifetime risk of complications soaring to 24%1. Arterial hypertension (AH) emerges as the most frequent cause or comorbidity linked with HF. Indeed, 80-90% of HF patients exhibit some form of AH, as hypertension-induced myocardial damage critically impairs heart function2. Therefore, the early detection of HF in individuals with hypertension is crucial. Utilizing biomarkers has proven to be an effective diagnostic approach. Of particular interest is the peptide sirtuin 1 (SIRT1), an enzyme protein that plays a key role in moderating apoptosis, energy metabolism, and influences life expectancy of cardiomyocytes by acting at both subcellular and epigenetic levels3, 4, 5. Research has linked SIRT1 expression with fibrotic processes in the heart and vascular wall6, 7, suggesting that plasma levels of this peptide could serve as indicators of myocardial dysfunction. The diagnostic potential of plasma SIRT1 levels for HF is supported by clinical studies, which show significant changes in patients with AH or those exhibiting clinical signs of HF8, 9, 10, 11. Yet, the data on its diagnostic utility among HF patients are mixed and sometimes contradictory, leaving several questions unanswered. Notably, factors such as age12, 13, and distinct effects of obesity across genders14, 15, can influence SIRT1 levels. Furthermore, the findings on plasma SIRT1 levels vary across different cardiovascular diseases (CVD), suggesting the need for targeted evaluation of plasma peptide levels in HF patients, considering potential genetic, age, gender, and anthropometric differences.
A crucial factor in a biomarker’s clinical application is understanding the genetic basis of its production. The gene controlling SIRT1 is located on chromosome 10q21.3 (ENSG00000096717). Research has explored several single nucleotide polymorphisms (SNPs) potentially impacting SIRT1 expression or structure: rs11599176, rs12413112, rs33957861, rs35689145, rs789600516, 17. Among these, the SNP with a cytosine to guanine substitution (C>G) at position rs7069102 stands out due to its significant impact on peptide expression and consequent plasma SIRT1 levels18. Studies reveal considerable variation in the prevalence of this SNP's variants across different populations (data sourced from the 1000 Genomes Project on https://www.ensembl.org), underscoring the importance of conducting region-specific research.
Given the multiple factors influencing plasma SIRT1 levels, we embarked on a cross-sectional study to delve into some of these aspects. The study’s objective was to explore potential anthropometric influences on plasma SIRT1 levels in hypertensive men with chronic HF, specifically considering the rs7069102 C>G polymorphism within the SIRT1 gene.
Methods
Patients
In planning our study groups, we drew inspiration from the designs of similar studies. Using the G*Power calculator (http://www.gpower.hhu.de/), we estimated a statistical power of 0.85 and a significance level of p < 0.05 to determine the minimum necessary group size. Accordingly, we enrolled 120 hypertensive men and 70 non-cardiovascular disease (CVD) men, who underwent examinations at the Vinnytsya Regional Diagnostic and Disease Prevention Center (VRDDP) in Vinnytsya, Ukraine, as our protocol dictates. The National Pirogov Memorial Medical University's (NPMMU) ethical committee, located in Vinnytsya, Ukraine, sanctioned our research protocol. Prior to commencing any procedure, informed consent was duly obtained from all participants who signed the required documents. We set the inclusion criteria to male individuals aged 40 to 65 years, residing in the Podil region of Ukraine, and diagnosed with essential arterial hypertension (EAH). Exclusion criteria were individuals with symptomatic arterial hypertension (AH), a history of myocardial infarction or unstable angina, significant congenital heart defects, arrhythmia, non-coronary myocardial diseases, pulmonary arterial hypertension, and severe kidney or liver dysfunction, alongside known rheumatological, endocrine, or oncological diseases. Data influencing our exclusion criteria included age, sex, and concurrent diseases' impact on plasma SIRT1 levels. The primary study group had an average age of 50.93 ± 0.449 years with an EAH diagnosis. Seventy control group participants showed no CVD signs, averaging 49.03 ± 0.794 years. Diagnoses followed the 2018 and 2021 European Society of Cardiology (ESC) guidelines, segregating 120 hypertensive men based on heart failure (HF) signs into two groups of 60: one showing no HF symptoms and the other displaying NYHA class II-III HF symptoms. Participants underwent extensive evaluations, including physicals, lab tests, blood pressure measurement, electrocardiography, echocardiography, and plasma SIRT1 level and SIRT1 gene rs7069102 C/G polymorphism analysis. Body mass index (BMI) calculations used the Quetelet formula (BMI = body weight (kg)/height2 (cm)), classifying obesity according to WHO guidelines.
Blood Samples
We assessed plasma SIRT1 levels and the rs7069102 SNP variant in the SIRT1 gene from venous blood samples of all participants. The SIRT1 plasma concentration was measured using the standard ELISA method with RayBiotech, Inc. (USA) reagents and a Humareader single enzyme analyzer (Germany). This choice was backed by ELISA's proven accuracy, specificity, and widespread clinical use.
Genomic DNA extracted from whole blood was used to determine the rs7069102 C/G polymorphism in the SIRT1 gene, employing the DNA-EXTRAN-1 reagent kit for extraction. The allele-specific polymerase chain reaction (PCR), a method renown for SNP analysis effectiveness, facilitated the detection of SIRT1 gene alleles at the rs7069102 position by tracking the fluorescent signal of the amplified products (Thermo Fisher Scientific, USA).
Statistical Analysis
We presented our findings as mean (M ± SEM) or percentages. Depending on the data distribution, we applied appropriate statistical methods to discern correlations or differences between groups. These included parametric (Pearson correlation, t-test) and non-parametric (Spearman correlation, Mann-Whitney U test) methods, one-way ANOVA with the Tukey criterion for parametric data, or the Kruskal–Wallis test for non-parametric data. We employed the χ2 Pearson criterion with Yates correction to assess differences in observation frequencies, especially for smaller group analyses. Differences were considered significant at p < 0.05.
| Parameters | Control group (n=70) | Group EAH without CHF (n=60) | Group EAH with CHF (n=60) |
|---|---|---|---|
| Age, years | 49.03±0.792 | 50.03±0.675 | 52.02±0.757 |
| Body weight, кg | 79.06±1.014 | 80.12±1.307 | 88.67±1.510 *# |
| BMI, кg/m 2 | 25.23±0.268 | 25.92±0.394 | 29.38±0.547 *# |
| GFR, ml/min/1.73 m² | 111.4±2.473 | 105.7±2.673 | 95.63±1.709 *# |
| Age of EAH onset, years | - | 48.98±1.119 | 35.77±1.120 # |
| Duration of EAH, years | - | 9.14±0.773 | 13.53±0.771 # |
| Obese persons, n (%) | 3 (4.29%) | 8 (13.34%) | 31 (57.67%) & |
| Groups | Control group (n = 70) | Group EAH without CHF (n = 60) | Group EAH with CHF (n = 60) |
|---|---|---|---|
| Individuals with variant СС+CG (n = 101) | 2.024±0.113 | 3.474±0.370 # | 1.744±0.126 & |
| Homozygotes GG (n = 89) | 1.668±0.160 | 3.091±0.308 # | 1.390±0.092 *& |
Results
Analysis of plasma SIRT1 concentrations in our study groups showed that men with essential arterial hypertension (EAH) not taking heart failure (HF) into account had significantly higher SIRT1 levels (2.414 ± 0.152 ng/ml) compared to the control group (1.891 ± 0.089 ng/ml, p = 0.014). Yet, among men with both EAH and HF, SIRT1 levels were notably lower (1.550±0.084 ng/ml) than in those with EAH alone (3.271 ± 0.238 ng/ml, p < 0.001) as illustrated in Figure 1.
Investigating further, we found a clear link between plasma peptide levels and either age or obesity. Upon reviewing the clinical characteristics, we confirmed all study groups were matched by age. However, differences in body weight and BMI were observed, particularly that individuals with both EAH and HF presented higher values compared to either men without any cardiovascular diseases (CVD) or those with EAH but no HF (Table 1).
Interestingly, the incidence of obesity was highest in the group suffering from both EAH and HF compared to either the control group or those with solely EAH. This group also displayed distinct hypertension profiles, characterized by earlier disease onset and prolonged duration, alongside notably lower glomerular filtration rates (GFR) than seen in other groups.
Subsequent correlation analyses between group differences and plasma SIRT1 levels uncovered no age correlation (R=-0.11, p=0.25). Nevertheless, significant correlations emerged regarding GFR (positive, R=+0.22, p=0.02), BMI (negative, R=-0.22, p=0.01), and disease duration (negative, R=-0.33, p<0.001). Exploration into the impact of moderate GFR reduction (60-90 ml/min) revealed no significant deviation in SIRT1 levels from those with normal renal function, suggesting SIRT1 plasma levels could serve as a reliable biomarker unaffected by age or renal status.
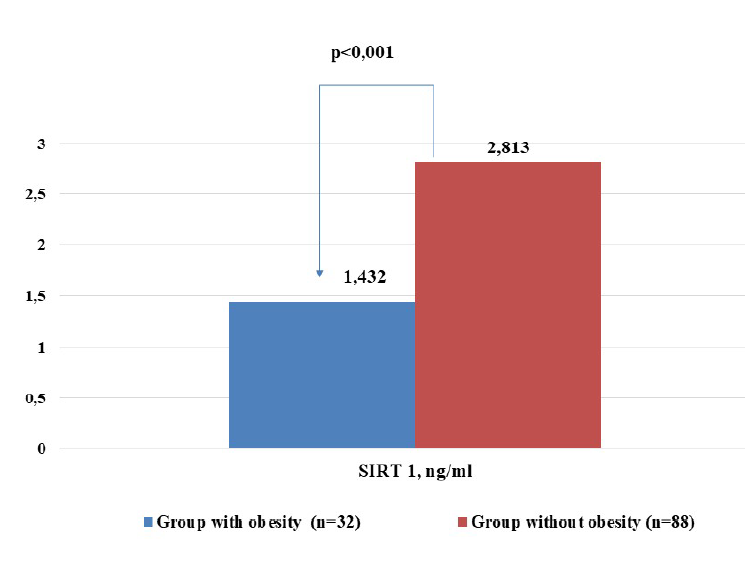
However, the presence of obesity drastically reduced plasma SIRT1 levels (1.432±0.117 ng/ml in obese men vs. 2.813±0.190 ng/ml in non-obese men, p<0.001) as shown in Figure 2. Early-onset EAH (up to age 35) was associated with lower SIRT1 levels (1.842±0.172 ng/ml) compared to later onset (2.665±0.187 ng/ml, p=0.009). These observations suggest that a prolonged history of hypertension and a higher prevalence of obesity might explain the decreased SIRT1 levels in patients with both EAH and HF.
Further analysis delved into the genetic influence on SIRT1 production, focusing on the rs7069102 SNP within the SIRT1 gene. Predominantly, in the Podil region of Ukraine, individuals carrying the mutant G allele were prevalent (70.26%). While CG and GG variants were equally distributed (both 46.84%), the CC homozygous variant was rare (6.32%). A detailed exploration of rs7069102 SNP variants revealed that, except for those with HF, SIRT1 plasma concentrations did not significantly differ among the groups, including between GG homozygotes and individuals carrying the CC or CG variants.
In conclusion, our research indicates that lower plasma SIRT1 levels in patients with both EAH and HF could be attributed to the interplay between disease duration, obesity, and specific genetic backgrounds. These findings underscore the complex nature of SIRT1 as a biomarker in hypertensive disorders, suggesting a need for more detailed studies to unravel the genetic underpinnings influencing its expression.
Discussion
We discovered that the plasma SIRT1 level in patients with EAH significantly differs from levels found in healthy volunteers. Research by Duman et al. suggests that in hypertensive patients, plasma peptide levels are notably higher than those in individuals with normal blood pressure19, 20, 21, 22. Conversely, other studies have indicated that hypertensive patients exhibit significantly lower plasma peptide concentrations compared to a control group9, 23. The assessment of SIRT1 in patients with heart failure (HF) showcases varied methodological approaches among researchers. Some studies have examined peptide expression levels in tissues, revealing experimental evidence that cardiomyocyte peptide expression in HF patients is nearly half that of healthy hearts8. Other analyses, focusing on SIRT1 expression in peripheral blood mononuclear cells, found significantly reduced levels in HF patients19. Despite the use of differing methodologies in these studies, our findings align closely. However, research conducted by Italian scientists reported higher peptide expression in HF patients than in healthy volunteers24. No studies were found examining plasma SIRT1 concentration in hypertensive patients with HF.
This diversity in results likely stems from the various study designs. When research includes participants of differing genders, ages, and HF etiologies, and employs varied methods for SIRT1 evaluation, interpreting or reproducing study findings becomes challenging. In planning our study, we minimized several variables by ensuring homogeneity in gender, age, and HF etiology within our groups. This approach allowed us to identify potential factors influencing plasma peptide levels: the duration of hypertension, disease severity, presence of obesity, and genetic components.
We observed that plasma SIRT1 concentration in patients with EAH and HF was significantly lower in those with early disease onset and obesity. This contrasts with findings in coronary heart disease (CHD) patients, where plasma peptide levels showed no correlation with anthropometric measures25, 26. Interestingly, Norwegian research identified a correlation between SIRT1 expression and obesity in women, but not in men14. The design of our cross-sectional study limits the generalization of our results, suggesting that men with EAH and HF may exhibit lower SIRT1 levels if obesity is present or if EAH appears early. This could impact the use of this peptide as a diagnostic marker for HF. However, our data supports a hypothesis that early EAH onset and obesity co-morbidity could accelerate HF development through more significant early hypertensive damage to the myocardium, depleting cardiomyocyte SIRT1 reserves. Experimental evidence links low SIRT1 expression in cardiomyocytes to advancing myocardial fibrosis27. Thus, reduced plasma SIRT1 levels could signal depleted myocardial functional reserves in HF development, meriting further longitudinal research.
We also explored the phenotypic significance of the rs7069102 C/G SNP in the SIRT1 gene concerning plasma activity and concentration levels. Minimal available literature exists on this topic. Kilic et al. found no significant differences in plasma peptide levels between carriers of different SNP variants in a general Turkish population12. Yet, in a CHD patient cohort, GG homozygotes exhibited significantly higher plasma SIRT1 levels compared to other variants28. In acute coronary syndrome patients, a higher plasma peptide concentration was linked to the C allele29. Little to no information exists on the rs7069102 C/G SNP's effect on plasma SIRT1 levels in HF patients. Our findings indicate low plasma peptide levels among homozygous GG variant carriers in patients with EAH and HF, offering new insights into the rs7069102 C/G polymorphism's phenotypic manifestations in Ukrainian hypertensive men. Given the minor allele G’s varying prevalence across populations, its potential impact on plasma SIRT1 levels could differ globally.
Our results lay the groundwork for further exploring plasma peptide concentrations in EAH patients, considering various heart structure and function disorders that lead to hypertensive heart and HF. The cross-sectional nature of our study precluded investigating other HF progression factors in EAH patients, such as lifestyle or treatment approaches. Consequently, our findings open avenues for detailed analysis of plasma SIRT1 level changes in patients with EH and varied HF phenotypes across different clinical scenarios. This could enhance the diagnostic and prognostic utility of SIRT1, refining the biomarker's application by accounting for significant influences and limitations.
Conclusions
In male patients diagnosed with exercise-associated hyponatremia (EAH), there was a significant association between lower plasma SIRT1 levels and heart failure (HF). Several factors contributing to reduced plasma peptide levels were identified, including obesity, the early onset of hypertension, and the presence of the GG variant within the rs7069102 single nucleotide polymorphism (SNP) in the SIRT1 gene. These findings enhance our understanding of SIRT1's diagnostic value and its potential limitations as a biomarker for HF in clinical settings. Furthermore, they underscore the necessity for and guide the direction of future research in this area.
Abbreviations
BMI - Body Mass Index, CHD - Coronary Heart Disease, CVD - Cardiovascular Disease, EAH - Essential Arterial Hypertension, ELISA - Enzyme-Linked Immunosorbent Assay, ESC - European Society of Cardiology, GFR - Glomerular Filtration Rate, HF - Heart Failure, NPMMU - National Pirogov Memorial Medical University, NYHA - New York Heart Association, PCR - Polymerase Chain Reaction, SEM - Standard Error of the Mean, SIRT1 - Sirtuin 1, SNP - Single Nucleotide Polorphism, VRDDP - Vinnytsya Regional Diagnostic and Disease Prevention Center, WHO - World Health Organization
Acknowledgments
This research received no specific grant from any funding agency in the public, commercial or non-profit sectors. We thank all patients for their participation in the investigation, and the doctors, nurses, and administrative staff of VRSRPD (Vinnytsia, Ukraine) that assisted with the study.
Author’s contributions
Starzhynska O.: developed data collection and assessment tools, analysis and interpretation of data, drafting of the manuscript. Donets A.: conducted the initial analysis of the problem, coordinated and supervised data collection on the site. Maiko O.: wrote review, edited of the manuscript. Zhebel V.: developed the study concept and design, edited and approved the final manuscript. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The research protocol was approved by the ethical committee of NPMM, Vinnytsya, Ukraine, which found no deviations from the requirements of the Declaration of Helsinki and the Council of Europe Convention on Human Rights and Biomedicine (1977) and requirements of the current legislation of Ukraine. Before initiating any procedures, all patients provided Informed Consent by signing the appropriate documentation.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
-
Bozkurt
B.,
Ahmad
T.,
Alexander
K.M.,
Baker
W.L.,
Bosak
K.,
Breathett
K.,
Writing Committee Members. Heart Failure Epidemiology and Outcomes Statistics: A Report of the Heart Failure Society of America. Journal of Cardiac Failure.
2023;
29
(10)
:
1412-1451
.
View Article PubMed Google Scholar -
Mills
K.T.,
Stefanescu
A.,
He
J.,
The global epidemiology of hypertension. Nature Reviews. Nephrology.
2020;
16
(4)
:
223-37
.
View Article PubMed Google Scholar -
Opstad
T.B.,
Berg
T.J.,
Holte
K.B.,
Arnesen
H.,
Solheim
S.,
Seljeflot
I.,
Reduced leukocyte telomere lengths and sirtuin 1 gene expression in long-term survivors of type 1 diabetes: A Dialong substudy. Journal of Diabetes Investigation.
2021;
12
(7)
:
1183-92
.
View Article PubMed Google Scholar -
Vargas-Ortiz
K.,
Pérez-Vázquez
V.,
Macías-Cervantes
M.H.,
Exercise and Sirtuins: A Way to Mitochondrial Health in Skeletal Muscle. International Journal of Molecular Sciences.
2019;
20
(11)
:
2717
.
View Article PubMed Google Scholar -
Wyman
A.E.,
Atamas
S.P.,
Sirtuins and Accelerated Aging in Scleroderma. Current Rheumatology Reports.
2018;
20
(4)
:
16
.
View Article PubMed Google Scholar -
Mengozzi
A.,
Costantino
S.,
Paneni
F.,
Duranti
E.,
Nannipieri
M.,
Mancini
R.,
Targeting SIRT1 Rescues Age- and Obesity-Induced Microvascular Dysfunction in Ex Vivo Human Vessels. Circulation Research.
2022;
131
(6)
:
476-91
.
View Article PubMed Google Scholar -
Paramesha
B.,
Anwar
M.S.,
Meghwani
H.,
Maulik
S.K.,
Arava
S.K.,
Banerjee
S.K.,
Sirt1 and Sirt3 Activation Improved Cardiac Function of Diabetic Rats via Modulation of Mitochondrial Function. Antioxidants.
2021;
10
(3)
:
338
.
View Article PubMed Google Scholar -
Lu
T.M.,
Tsai
J.Y.,
Chen
Y.C.,
Huang
C.Y.,
Hsu
H.L.,
Weng
C.F.,
Downregulation of Sirt1 as aging change in advanced heart failure. Journal of Biomedical Science.
2014;
21
(1)
:
57
.
View Article PubMed Google Scholar -
Martinez-Arroyo
O.,
Ortega
A.,
Galera
M.,
Solaz
E.,
Martinez-Hervas
S.,
Redon
J.,
Decreased Urinary Levels of SIRT1 as Non-Invasive Biomarker of Early Renal Damage in Hypertension. International Journal of Molecular Sciences.
2020;
21
(17)
:
6390
.
View Article PubMed Google Scholar -
Waldman
M.,
Cohen
K.,
Yadin
D.,
Nudelman
V.,
Gorfil
D.,
Laniado-Schwartzman
M.,
Regulation of diabetic cardiomyopathy by caloric restriction is mediated by intracellular signaling pathways involving `SIRT1 and PGC-1α'. Cardiovascular Diabetology.
2018;
17
(1)
:
111
.
View Article PubMed Google Scholar -
Yan
J.,
Wang
J.,
He
J.C.,
Zhong
Y.,
Sirtuin 1 in Chronic Kidney Disease and Therapeutic Potential of Targeting Sirtuin 1. Frontiers in Endocrinology (Lausanne).
2022;
13
.
View Article PubMed Google Scholar -
Kilic
U.,
Gok
O.,
Elibol-Can
B.,
Uysal
O.,
Bacaksiz
A.,
Efficacy of statins on sirtuin 1 and endothelial nitric oxide synthase expression: the role of sirtuin 1 gene variants in human coronary atherosclerosis. Clinical and Experimental Pharmacology & Physiology.
2015;
42
(4)
:
321-30
.
View Article PubMed Google Scholar -
Zhong
Y.,
Chen
A.F.,
Zhao
J.,
Gu
Y.J.,
Fu
G.X.,
Serum levels of cathepsin D, sirtuin1, and endothelial nitric oxide synthase are correlatively reduced in elderly healthy people. Aging Clinical and Experimental Research.
2016;
28
(4)
:
641-5
.
View Article PubMed Google Scholar -
Opstad
T.B.,
Kalstad
A.A.,
Pettersen
A.\AA.,
Arnesen
H.,
Seljeflot
I.,
Novel biomolecules of ageing, sex differences and potential underlying mechanisms of telomere shortening in coronary artery disease. Experimental Gerontology.
2019;
119
:
53-60
.
View Article PubMed Google Scholar -
Doulamis
I.P.,
Tzani
A.I.,
Konstantopoulos
P.S.,
Samanidis
G.,
Georgiopoulos
G.,
Toutouzas
K.P.,
A sirtuin 1/MMP2 prognostic index for myocardial infarction in patients with advanced coronary artery disease. International Journal of Cardiology.
2017;
230
:
447-53
.
View Article PubMed Google Scholar -
Clark
S.J.,
Falchi
M.,
Olsson
B.,
Jacobson
P.,
Cauchi
S.,
Balkau
B.,
Association of sirtuin 1 (SIRT1) gene SNPs and transcript expression levels with severe obesity. Obesity (Silver Spring, Md.).
2012;
20
(1)
:
178-85
.
View Article PubMed Google Scholar -
Dardano
A.,
Lucchesi
D.,
Garofolo
M.,
Gualdani
E.,
Falcetta
P.,
Sancho Bornez
V.,
SIRT1 rs7896005 polymorphism affects major vascular outcomes, not all-cause mortality, in Caucasians with type 2 diabetes: A 13-year observational study. Diabetes/Metabolism Research and Reviews.
2022;
38
(4)
.
View Article PubMed Google Scholar -
Yamac
A.H.,
Uysal
O.,
Ismailoglu
Z.,
Ertürk
M.,
Celikten
M.,
Bacaksiz
A.,
Premature Myocardial Infarction: Genetic Variations in SIRT1 Affect Disease Susceptibility. Cardiology Research and Practice.
2019;
2019
.
View Article PubMed Google Scholar -
Akkafa
F.,
Halil Altiparmak
I.,
Erkus
M.E.,
Aksoy
N.,
Kaya
C.,
Ozer
A.,
Reduced SIRT1 expression correlates with enhanced oxidative stress in compensated and decompensated heart failure. Redox Biology.
2015;
6
:
169-73
.
View Article PubMed Google Scholar -
Conti
V.,
Corbi
G.,
Manzo
V.,
Malangone
P.,
Vitale
C.,
Maglio
A.,
SIRT1 Activity in Peripheral Blood Mononuclear Cells Correlates with Altered Lung Function in Patients with Chronic Obstructive Pulmonary Disease. Oxidative Medicine and Cellular Longevity.
2018;
2018
.
View Article PubMed Google Scholar -
Starzhynska
O.,
Donets
A.,
Prevalence of the single nucleotide polymorphism (sSNP) rs7069102 of the SIRT1 gene among hypertensive residents of the Podil region of Ukraine. Precarpathian bulletin of the Shevchenko scientific society. Pulse..
2023;
19
(67)
:
7-18
.
View Article Google Scholar -
Duman
H.,
Bahçeci
I.,
Çinier
G.,
Duman
H.,
Bak\irc\i
E.M.,
Çetin
M.,
Left ventricular hypertrophy is associated with increased sirtuin level in newly diagnosed hypertensive patients. Clinical and Experimental Hypertension (New York, N.Y.).
2019;
41
(6)
:
511-5
.
View Article PubMed Google Scholar -
Prola
A.,
Pires Da Silva
J.,
Guilbert
A.,
Lecru
L.,
Piquereau
J.,
Ribeiro
M.,
SIRT1 protects the heart from ER stress-induced cell death through eIF2α deacetylation. Cell Death and Differentiation.
2017;
24
(2)
:
343-56
.
View Article PubMed Google Scholar -
Corbi
G.,
Conti
V.,
Troisi
J.,
Colucci
A.,
Manzo
V.,
Di Pietro
P.,
Cardiac Rehabilitation Increases SIRT1 Activity and β-Hydroxybutyrate Levels and Decreases Oxidative Stress in Patients with HF with Preserved Ejection Fraction. Oxidative Medicine and Cellular Longevity.
2019;
2019
.
View Article PubMed Google Scholar -
Samant
S.A.,
Pillai
V.B.,
Gupta
M.P.,
Cellular mechanisms promoting cachexia and how they are opposed by sirtuins. Canadian Journal of Physiology and Pharmacology.
2019;
97
(4)
:
235-45
.
View Article PubMed Google Scholar -
Yang
P.,
Dai
T.,
Liu
B.,
Huang
L.,
Yin
J.,
Zhao
F.,
The Predictive Value of NGF, TMAO, SIRT1 and apoA1 in Patients With Ischemic Heart Failure. Alternative Therapies in Health and Medicine.
2023;
2023
:
AT9328
.
PubMed Google Scholar -
Liu
Y.P.,
Wen
R.,
Liu
C.F.,
Zhang
T.N.,
Yang
N.,
Cellular and molecular biology of sirtuins in cardiovascular disease. Biomedicine and Pharmacotherapy.
2023;
164
.
View Article PubMed Google Scholar -
Kilic
U.,
Gok
O.,
Bacaksiz
A.,
Izmirli
M.,
Elibol-Can
B.,
Uysal
O.,
SIRT1 gene polymorphisms affect the protein expression in cardiovascular diseases. PLoS One.
2014;
9
(2)
.
View Article PubMed Google Scholar -
Kızıltunç
E.,
Kösem
A.,
Özkan
C.,
Ilgın
B.U.,
Kundi
H.,
Çetin
M.,
Serum Sirtuin 1, 3 and 6 Levels in Acute Myocardial Infarction Patients. Arquivos Brasileiros de Cardiologia.
2019;
113
(1)
:
33-9
.
View Article PubMed Google Scholar
Comments
Article Details
Volume & Issue : Vol 11 No 4 (2024)
Page No.: 6289-6296
Published on: 2024-04-30
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 3508 times
- PDF downloaded - 979 times
- XML downloaded - 124 times
 Biomedpress
Biomedpress




